
This week we're going behind the scenes of the most fascinating, and honestly underrated, corner of regulatory affairs: medical devices.
We're breaking down the terms you need to know, spotlighting some truly spectacular industry oopsies, and catching up on the latest in health, tech, and wellness. Let's get into it.
A Cheat Sheet for People Who Think Medical Devices Are Boring
Dude, I have to tell you something:
Medical devices are the most interesting category in all of regulatory affairs, and the fact that nobody talks about them like that is asinine (I watch too much King of the Hill).
We're talking about AI-powered implants, robotic surgery systems, pacemakers that connect to the internet, and tiny sensors that live inside your blood vessels. Meanwhile, RA professionals are out here explaining them with the energy of someone reading a tax form.
Let's fix that. Here's your crash course in medical device terminology: explained like a human being.
Class I: The "Probably Fine" Tier ✦ About 47% of all devices are Class I. FDA mostly leaves them alone — which is also why 10.8% of 2024 recalls were Class I devices. Complacency is a real thing. |
Class II: The “Middle Child” Tier ✦ This is the most common clearance tier. If you work in medtech, you will live in Class II territory for most of your career. |
Class III: The “Please Don’t Mess This Up” Tier |
510(K): The “Device Has a Cousin” Pathway |
The thing about medical devices is that they fail pretty publicly. Unlike a supplement that doesn’t (actually) work, a device failure tends to show up in data, in MDRs, in 911 cals, in lawsuits. That accountability is a feature. It’s also why this category demands the most rigorous RA work you will every do.
Big Oopsies
The Hall of Shame: Medical Device Edition
2024 was a record year for medical device recalls: 1059 events, a four-year high. Device failure was the leading cause for the first time in over five years. So let’s talk about some of the biggest, most instructive, and most spectacular failures in recent memory.
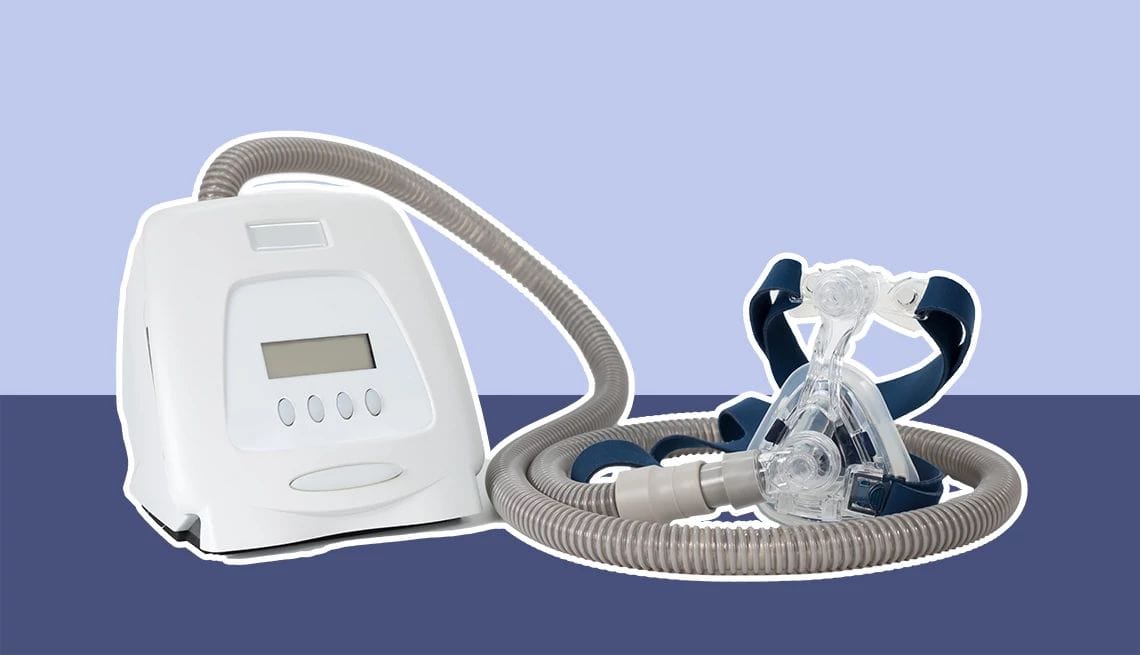
Biggest Oopsie Of All Time
Oopsie #1 Philips Respironics: The Foam That Poisoned Millions of Sleepers
- What happened: Philips built sound-dampening foam into its CPAP, BiPAP, and ventilator machines to reduce noise. A reasonable idea. Except the foam, a polyester-based polyurethane (PE-PUR), degraded over time, especially in warm, humid environments (you know, like the inside of a breathing machine that sits in your bedroom). The foam broke down into particles and toxic gases, including formaldehyde, which users were then inhaling while they slept. Every night. For years.
- The scale: The recall covered roughly 15 million devices sold between 2008 and 2021. FDA received over 116,000 Medical Device Reports, including 561 deaths, connected to the foam degradation. Philips knew there were complaints as early as 2015 and didn't issue the recall until June 2021.
- The aftermath: Philips agreed to a $1.1 billion personal injury settlement, a separate $479 million class-action settlement for economic losses, and a consent decree with FDA and the DOJ that banned them from selling new CPAP and BiPAP devices in the US until conditions were met. They stopped selling entirely. Philips, one of the most trusted names in healthcare, effectively exited the US sleep apnea market they had dominated for years.Oopsie #2 Life-Saving Devices Are Also The Most-Recalled Devices.
- What happened: A 2024–2025 analysis of FDA recall data found that the three most frequently recalled device categories were ventilators, resuscitators, and infusion pumps: all Class I recalls, meaning the most serious type, with potential for serious injury or death. Philips topped the manufacturer list with 5 recalls affecting 383,141 units. Smiths Medical came in second with 4 recalls affecting nearly 1 million units.
- Why this matters: These aren't fringe products. Ventilators keep people alive when they can't breathe. Infusion pumps deliver precise medication doses. Resuscitators restart hearts. The fact that these categories dominate the recall list isn't a coincidence, they're complex, used in high-stakes environments, and often aging in hospital storage rooms for years between maintenance cycles.Oopsie #3 STRYKER - When A Medical Device Gets Wiped Out by Hackers
- What happened: On March 11, 2026, an Iran-linked hacking group called Handala launched a cyberattack on Stryker, a Fortune 500 medical device maker that makes everything from orthopedic implants to defibrillators. The attackers claimed to have wiped more than 200,000 devices, servers, and laptops across 79 countries. They used Stryker's own Microsoft Intune device management system to remotely wipe thousands of employee devices. The attack claimed to be retaliation for US military action in Iran.
- The impact: Order processing, manufacturing, and shipping ground to a halt. Surgeries were rescheduled because custom implant orders couldn't be fulfilled. Stryker's LIFENET system, used by emergency responders to transmit ECG data to hospitals, went temporarily offline in parts of Maryland. CISA issued a national alert urging healthcare organizations to harden their endpoint security in direct response to the Stryker attack.
- What Stryker said: The devices themselves were safe. No ransomware. The attack was contained to internal IT systems. Stryker filed an 8-K with the SEC and brought in Palo Alto Networks Unit 42 to investigate. Estimated device restoration cost alone: $24–40 million, before you count lost productivity, incident response, or delayed surgeries.Oopsie #4 AI Medical Devices: Cleared Fast, Recalled Faster
- What happened: A Johns Hopkins study examined 950 AI-enabled medical devices authorized by FDA through November 2024. Of those, 60 devices were associated with 182 recall events. The most common causes: diagnostic errors and functionality failures. The most alarming finding: 43% of all recalls happened within one year of FDA clearance. The majority of recalled AI devices had not gone through clinical trials before reaching the market.
- Why: Most AI devices reach the market through the 510(k) pathway, which doesn't require clinical trials. If the algorithm is deemed "substantially equivalent" to a predicate, it can get cleared without prospective human testing. That works fine for a lot of devices. For AI tools making diagnostic decisions? The gap between lab performance and real-world performance can be brutal.
- Publicly traded companies accounted for 53% of recalled AI devices and 98.7% of recalled units. Researcher Tinglong Dai from Johns Hopkins attributed this to investor-driven pressure for faster launches.
In case you missed it
The Tea
The latest news on tech, health & wellness, and clinical research.

OnlyFans billionaire funded clinical trials
OnlyFans Billionaire’s Cancer Fight. Leonid Radvinsky died at 43 from cancer despite $23M in self-funded research grants and MSK donations. His wealth bought top care but couldn't shortcut clinical trials' validation process. Even billionaires need the research infrastructure working for breakthroughs. Perfect case study for why RA pros protect trial integrity: no exceptions.
Health tech founder cashes out $700M EHR exit to tackle U.S. shelters: 6.5M animals enter yearly, 920K euthanized. Ryan Howard is building AI for lifestyle-based pet matching and free tools for cash-strapped rescues. The pivot proves mission trumps money when systems fail animals at this scale. Shows health tech lessons (data + AI) can solve adjacent crises.
Invisible Diagnostics Are Health’s Next Big Thing. The Global Wellness Summit's March 2026 trend report declared we're entering the era of "ambient health intelligence." Smart toilets (Throne) analyzing your gut health. Microsensors on your teeth (Lura) tracking glucose and biologics. Tampons (Comma) running blood tests for reproductive health. The theme: diagnostics you don't have to think about. The regulatory reality: FDA is going to have a lot of opinions about teeth sensors and smart toilets that claim to flag cancer risk.
Utah Becomes The First State To Allow AI To Autonomously Review Prescriptions. Utah passed legislation allowing AI systems to autonomously renew certain prescription drugs without direct physician involvement. It's a narrow carve-out, not a free-for-all, but it's the first legislative crack in a wall that has held for decades. The downstream regulatory implications for prescribing, liability, and the definition of "practice of medicine" are going to be debated for years.
Tech moves fast, but you're still playing catch-up?
That's exactly why 200K+ engineers working at Google, Meta, and Apple read The Code twice a week.
Here's what you get:
Curated tech news that shapes your career - Filtered from thousands of sources so you know what's coming 6 months early.
Practical resources you can use immediately - Real tutorials and tools that solve actual engineering problems.
Research papers and insights decoded - We break down complex tech so you understand what matters.
All delivered twice a week in just 2 short emails.
That’s it for this week
Why is nobody talking about this?
Silicon Valley gave us a philosophy: ship it, learn from failures, iterate. It works beeeauutifully for apps. It is, however, a patient safety catastrophe when applied to medical devices.
“The companies that kill it aren’t the ones that ignore regulations. They’re the ones that understand them well enough to push the boundary without falling off.”
We have companies moving too fast to market (43% of AI devices recalled within one year). An FDA that is under-resourced to keep up: the GAO reported in December 2025 that FDA couldn't meet its three-month recall closure goal because of a hiring freeze. And a healthcare system that depends on both of them to get it right.
Meanwhile, Utah just passed a law letting AI autonomously renew prescriptions. OpenAI launched a consumer health tool. And pro-Iranian hackers wiped 200,000 devices at a Fortune 500 medical device company as a geopolitical message.
Medical devices have never been higher-stakes. Or more in need of people who actually understand how the regulatory system works.
That's you. Do not undersell yourself at dinner parties. 🖤
Like what you see? Share with a friend, colleague or frenemy.
Until next week,
Kristina



